| Category | |
| Keywords | Industrial Enzymes |
| Current development stage | General list: TRL3 Experimental proof of concept |
| Collaboration Opportunity | Sponsored Research with an option to License Research Results |
Background
Lipase enzymes are a subclass of the esterases. They perform essential roles in digestion, transport, and processing of dietary lipids in living organisms. Under natural conditions, lipases catalyze the hydrolysis of ester bonds at the interface between an insoluble substrate phase and the aqueous phase where the enzyme remains dissolved.
Lipase enzymes are the most important group of biocatalysts for biotechnological applications, and they are used in a wide array of industrial scale applications, such as: synthesis of biopolymers, the production of biodiesel, enantiopure pharmaceuticals, agrochemicals, biosensors and flavor compounds. Many of these industrial scale synthetic reactions are carried out in organic solvents because of the easy solubility of non-polar compounds. However, although enzymatic reactions in organic media show numerous advantages, the catalytic efficiency of most enzymes is usually orders of magnitude lower than in aqueous systems. Unlike lipases from plants and animals, some microbial lipases tend to be stable in organic solvents. Together with other desired properties such as thermostability, they have great potential as industrial enzymes.
Our Innovation
Dr. Levy identified a fungi lipase that is stable at extremely high temperatures and has a very high tolerance to organic solvents.
Technology
Our results show that our fungi isolate produces extremely thermostable lipases that have optimal activity temperature of about 90⁰C (Figure 1). Very few lipases have been reported in scientific literature which exhibit optimal activity at such high temperatures. In addition to the thermo-stability, this lipase was also found to have high tolerance to some organic solvents.
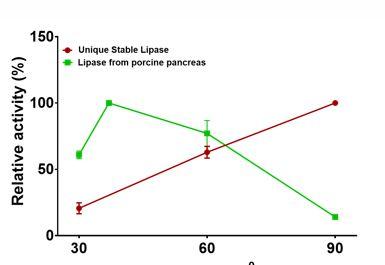
Figure 1. Temperature Effect on Lipase Activity
This fungi lipase was tested in various organic solvents. Results, in Figure 2 below, demonstrate:
- That 25% DMSO had a very small negative effect on the activity of this lipase, and the relative activity was 97% in comparison to the activity in aqueous buffer. At 50% DMSO[LG8] [IP9] [LG10] , the relative activity was reduced to 72%.
- At 25% isopropanol, the lipase activity actually increased to 133% when compared with that of an aqueous buffer. When the concentration of isopropanol reached 50%, the relative activity decreased to 47%.
- At 25% methanol, the lipase activity increased to over 200%.
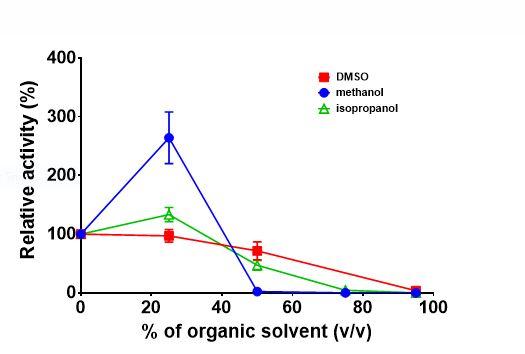
Figure 2. Lipase Activity in Different Solvents
Opportunity
Dr. Levy is looking for industry collaboration, sponsored research and licensing of the above mentioned thermostable and organo-solvent stable lipase.

