| Category | Diagnostic, Food Tech Vet Tech |
| Keywords | Spore Identification |
| Current development stage | General list: TRL3 Experimental proof of concept |
| Collaboration Opportunity | Licensing of Technology |
Background
Bacteria enter hibernation, becoming spores, when environmental conditions are sub optimal. Spores are exceptionally hardy and can withstand extreme heat, radiation and chemical assault for extended periods of time. Bacterial spores have even been found to be over one million years old.
Bacterial spores are troublesome for various industries but particularly in the medical and food industries as they withstand traditional sanitation techniques, such as pasteurization and insufficiently harsh autoclaves. There are two types of spore sensing kits, mail-in and in-office, that are currently available in the marketplace to test the sanitation of the tools (equipment, implants, media) in case of autoclaves failure. However, these kit options need an incubation time of 24 minutes to 24 hours (depending on the kit type) to determine if the sanitation procedure was sufficient to kill spores. There are other limitations in terms of efficiency, accuracy, and sensitivity.
Our Innovation
The researchers have discovered a method that can reduce the incubation time of in-office sanitation kits from hours to mere minutes. Through the researchers’ investigations they were able to determine that spores transition from hibernation to revival occurs within five minutes, and their novel method can identify this transition by simple spectroscopy.
Technology
The technology is based on a genetically modified B. subtilis that contains a reporter gene which is expressed immediately upon revival of the spores. These reporter genes can either be green-fluorescent protein (GFP) or the more common beta -Glucuronidase, which can be identified in a simple fluorometric assay.
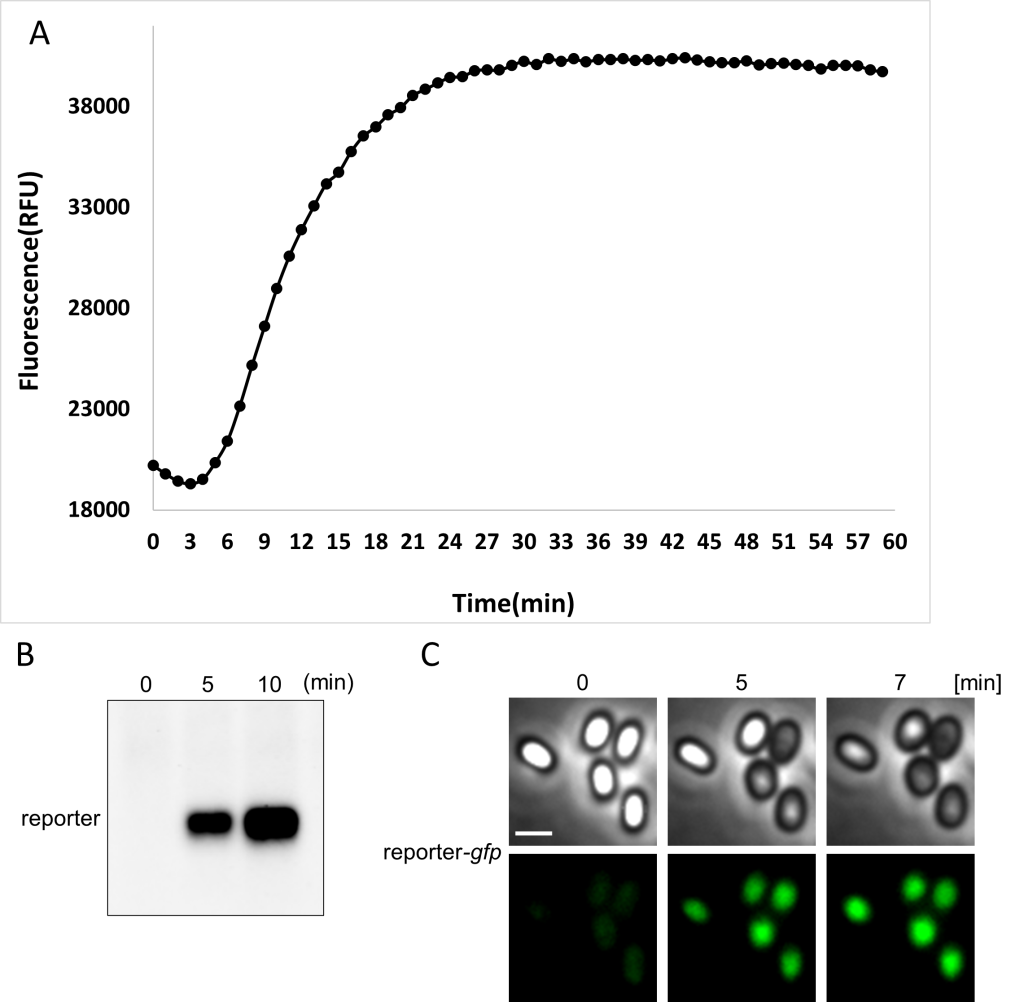
In the below figure, genetically modified spores containing a GFP reporter were woken and analyzed by A) fluorometer B) western blot, and C) microscopy. After five minutes it is possible to recognize a fluorescent signal, indicating that the spores are alive.
Opportunity
We are looking for an industrial partner to take a license to the described technology, and to develop a rapid spore testing bioassay kit that can be used on-site to get rapid confirmation of autoclave efficiency.
